Strengthening laboratory systems to enhance patient outcomes in Angola
-
by
AFENET
During 2019-2024, AFENET supported Angola as part of broader efforts to strengthen laboratory systems and improve the national response to HIV and TB. Working under the PEPFAR/CDC-supported program, AFENET worked through strategic partnerships with the Ministry of Health (MoH), Instituto Nacional de Luta contra a SIDA (INLS), Instituto Nacional de Investigação em Saúde (INIS), PEPFAR/CDC International Laboratory Branch (ILB), provincial health directorates in Luanda, Benguela, Cunene, Lunda Sul, and Huambo, SystemOne, private sample transport networks, and facility-based laboratory teams to address longstanding gaps in diagnostic accuracy, quality assurance, workforce capacity, and sample referral networks. Prior to these interventions, many facilities faced challenges such as limited testing coverage, inconsistent quality standards, weak data systems, and delayed results—all of which hindered timely diagnosis and treatment.
 AFENET significantly strengthened HIV rapid testing quality by supporting the national HIV Rapid Testing External Quality Assessment (EQA) program through the preparation, distribution, and evaluation of proficiency testing panels across five provinces— Luanda, Benguela, Huambo, Cunene, and Lunda Sul. We trained and supervised HIV testers, conducted corrective action follow-ups, and, for the first time, mapped HIV testers across 22 health facilities. We also led the national adoption and rollout of the three-test HIV screening algorithm, contributed to the development of national guidelines, and introduced standardized training and logbook tools. A major milestone was developing and piloting Angola’s first HIV Rapid Test Certification curriculum, through which 46 testers were initially trained.
To expand and improve TB diagnostic capacity, we provided EQA/PT support for Acid-Fast Bacillus testing and the GeneXpert MTB/RIF program, increasing the number of participating facilities from 10 to 15 across four provinces. We also supported the standardization of logbooks, strengthened quality control processes, and facilitated the initiation of both Xpert and AFB testing services in multiple health facilities. Quality Management Systems (QMS) and continuous improvement efforts were strengthened through the rollout of a comprehensive QMS package consisting of more than 100 tools, coupled with ongoing mentorship and supportive supervision in over 22 health facilities. These efforts contributed to improved SIMS (Site Improvement Through Monitoring System) scores in the supported laboratories.
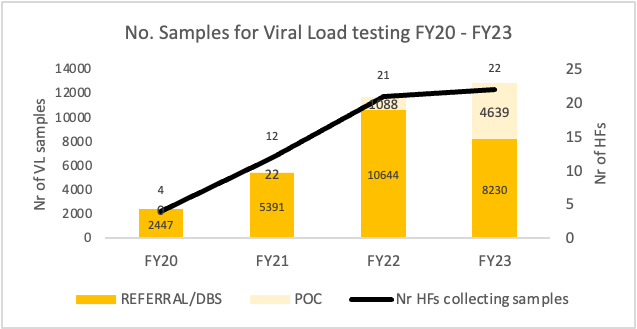
To enhance sample transport and laboratory information systems, AFENET developed SOPs and trained couriers to reinforce VL/EID sample transport mechanisms. We contracted private transport companies and leveraged national networks to ensure efficient transport, including to remote provinces. Additionally, the Appolo LIS was deployed in both Luanda and Benguela provinces, and qualified staff were seconded to enhance performance, improving data availability, reporting accuracy, and turnaround time monitoring across the laboratory network.
AFENET’s support in Angola greatly strengthened the country’s HIV and TB diagnostic landscape by improving quality assurance systems, increasing access to reliable testing, enhancing data use, and building sustainable laboratory capacity. Through technical mentorship, innovations in diagnostic quality control, and strong multisectoral strategic partnerships, Angola is now better equipped to ensure accurate diagnosis, guide treatment decisions, and advance toward epidemic control. The success of these interventions demonstrates how targeted laboratory system strengthening can drive measurable public health gains and lay a strong foundation for future disease surveillance and response.
AFENET significantly strengthened HIV rapid testing quality by supporting the national HIV Rapid Testing External Quality Assessment (EQA) program through the preparation, distribution, and evaluation of proficiency testing panels across five provinces— Luanda, Benguela, Huambo, Cunene, and Lunda Sul. We trained and supervised HIV testers, conducted corrective action follow-ups, and, for the first time, mapped HIV testers across 22 health facilities. We also led the national adoption and rollout of the three-test HIV screening algorithm, contributed to the development of national guidelines, and introduced standardized training and logbook tools. A major milestone was developing and piloting Angola’s first HIV Rapid Test Certification curriculum, through which 46 testers were initially trained.
To expand and improve TB diagnostic capacity, we provided EQA/PT support for Acid-Fast Bacillus testing and the GeneXpert MTB/RIF program, increasing the number of participating facilities from 10 to 15 across four provinces. We also supported the standardization of logbooks, strengthened quality control processes, and facilitated the initiation of both Xpert and AFB testing services in multiple health facilities. Quality Management Systems (QMS) and continuous improvement efforts were strengthened through the rollout of a comprehensive QMS package consisting of more than 100 tools, coupled with ongoing mentorship and supportive supervision in over 22 health facilities. These efforts contributed to improved SIMS (Site Improvement Through Monitoring System) scores in the supported laboratories.
To enhance sample transport and laboratory information systems, AFENET developed SOPs and trained couriers to reinforce VL/EID sample transport mechanisms. We contracted private transport companies and leveraged national networks to ensure efficient transport, including to remote provinces. Additionally, the Appolo LIS was deployed in both Luanda and Benguela provinces, and qualified staff were seconded to enhance performance, improving data availability, reporting accuracy, and turnaround time monitoring across the laboratory network.
AFENET’s support in Angola greatly strengthened the country’s HIV and TB diagnostic landscape by improving quality assurance systems, increasing access to reliable testing, enhancing data use, and building sustainable laboratory capacity. Through technical mentorship, innovations in diagnostic quality control, and strong multisectoral strategic partnerships, Angola is now better equipped to ensure accurate diagnosis, guide treatment decisions, and advance toward epidemic control. The success of these interventions demonstrates how targeted laboratory system strengthening can drive measurable public health gains and lay a strong foundation for future disease surveillance and response.
 AFENET significantly strengthened HIV rapid testing quality by supporting the national HIV Rapid Testing External Quality Assessment (EQA) program through the preparation, distribution, and evaluation of proficiency testing panels across five provinces— Luanda, Benguela, Huambo, Cunene, and Lunda Sul. We trained and supervised HIV testers, conducted corrective action follow-ups, and, for the first time, mapped HIV testers across 22 health facilities. We also led the national adoption and rollout of the three-test HIV screening algorithm, contributed to the development of national guidelines, and introduced standardized training and logbook tools. A major milestone was developing and piloting Angola’s first HIV Rapid Test Certification curriculum, through which 46 testers were initially trained.
To expand and improve TB diagnostic capacity, we provided EQA/PT support for Acid-Fast Bacillus testing and the GeneXpert MTB/RIF program, increasing the number of participating facilities from 10 to 15 across four provinces. We also supported the standardization of logbooks, strengthened quality control processes, and facilitated the initiation of both Xpert and AFB testing services in multiple health facilities. Quality Management Systems (QMS) and continuous improvement efforts were strengthened through the rollout of a comprehensive QMS package consisting of more than 100 tools, coupled with ongoing mentorship and supportive supervision in over 22 health facilities. These efforts contributed to improved SIMS (Site Improvement Through Monitoring System) scores in the supported laboratories.
To enhance sample transport and laboratory information systems, AFENET developed SOPs and trained couriers to reinforce VL/EID sample transport mechanisms. We contracted private transport companies and leveraged national networks to ensure efficient transport, including to remote provinces. Additionally, the Appolo LIS was deployed in both Luanda and Benguela provinces, and qualified staff were seconded to enhance performance, improving data availability, reporting accuracy, and turnaround time monitoring across the laboratory network.
AFENET’s support in Angola greatly strengthened the country’s HIV and TB diagnostic landscape by improving quality assurance systems, increasing access to reliable testing, enhancing data use, and building sustainable laboratory capacity. Through technical mentorship, innovations in diagnostic quality control, and strong multisectoral strategic partnerships, Angola is now better equipped to ensure accurate diagnosis, guide treatment decisions, and advance toward epidemic control. The success of these interventions demonstrates how targeted laboratory system strengthening can drive measurable public health gains and lay a strong foundation for future disease surveillance and response.
AFENET significantly strengthened HIV rapid testing quality by supporting the national HIV Rapid Testing External Quality Assessment (EQA) program through the preparation, distribution, and evaluation of proficiency testing panels across five provinces— Luanda, Benguela, Huambo, Cunene, and Lunda Sul. We trained and supervised HIV testers, conducted corrective action follow-ups, and, for the first time, mapped HIV testers across 22 health facilities. We also led the national adoption and rollout of the three-test HIV screening algorithm, contributed to the development of national guidelines, and introduced standardized training and logbook tools. A major milestone was developing and piloting Angola’s first HIV Rapid Test Certification curriculum, through which 46 testers were initially trained.
To expand and improve TB diagnostic capacity, we provided EQA/PT support for Acid-Fast Bacillus testing and the GeneXpert MTB/RIF program, increasing the number of participating facilities from 10 to 15 across four provinces. We also supported the standardization of logbooks, strengthened quality control processes, and facilitated the initiation of both Xpert and AFB testing services in multiple health facilities. Quality Management Systems (QMS) and continuous improvement efforts were strengthened through the rollout of a comprehensive QMS package consisting of more than 100 tools, coupled with ongoing mentorship and supportive supervision in over 22 health facilities. These efforts contributed to improved SIMS (Site Improvement Through Monitoring System) scores in the supported laboratories.
To enhance sample transport and laboratory information systems, AFENET developed SOPs and trained couriers to reinforce VL/EID sample transport mechanisms. We contracted private transport companies and leveraged national networks to ensure efficient transport, including to remote provinces. Additionally, the Appolo LIS was deployed in both Luanda and Benguela provinces, and qualified staff were seconded to enhance performance, improving data availability, reporting accuracy, and turnaround time monitoring across the laboratory network.
AFENET’s support in Angola greatly strengthened the country’s HIV and TB diagnostic landscape by improving quality assurance systems, increasing access to reliable testing, enhancing data use, and building sustainable laboratory capacity. Through technical mentorship, innovations in diagnostic quality control, and strong multisectoral strategic partnerships, Angola is now better equipped to ensure accurate diagnosis, guide treatment decisions, and advance toward epidemic control. The success of these interventions demonstrates how targeted laboratory system strengthening can drive measurable public health gains and lay a strong foundation for future disease surveillance and response. 


