AFENET strengthens HIV care and laboratory systems in The Dominican Republic
-
by
AFENET
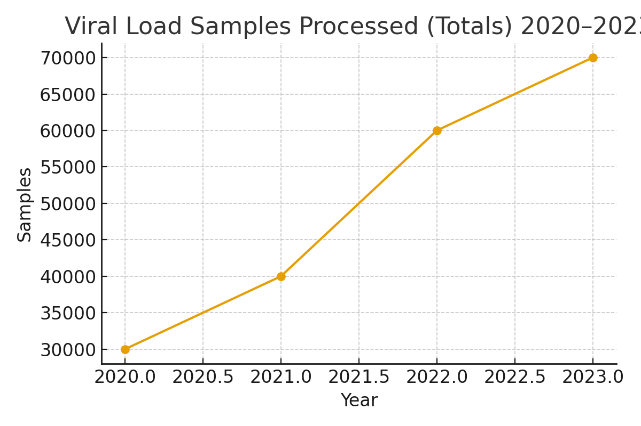
AFENET was the only PEPFAR implementing partner with nationwide reach in the Dominican Republic during the period 2020 – 2023. Working in close partnership with the Ministry of Health, the National Health Service (SNS), CDC/PEPFAR and other implementing partners, we expanded access to high-quality care across all 32 provinces by building an integrated network of clinical and laboratory services aligned with international standards. We also introduced advanced viral load and rapid testing for recent HIV infection. These interventions resulted in improved HIV care and laboratory systems in all
74 comprehensive HIV Care Services (SAI’s) and PEPFAR’s 39 prioritized SAI’s along with their respective laboratories.
More specifically, we were able to achieve the following:
- 650,000 HIV rapid tests performed, detecting 26,000 HIV-positive individuals, and consistently achieving the first 95% target.
- 44,000 people started on ART in 2023, compared to 39,500 people in 2021
- Markedly improved viral suppression from about 25,000 people (63% of those on ART and 41% of all diagnosed) in 2021 to 46,000 (88 % of those on ART and 56 % of all diagnosed) in 2023.
- Guided the Dr. Defilló National Public Health Reference Laboratory to ISO 15189:2022 accreditation, with promising results.
- Conducted three SLMTA (Strengthening Laboratory Management Toward Accreditation) workshops and certified 22 internal auditors, 4 national facilitators, and 1 master trainer.
- 150 laboratory staff trained in sample management and biosafety
- Engaged more than 100 participants in rapid HIV test quality assurance; undertook several External Quality Assessment and DNA-PCR infant diagnosis workshops for 260 key personnel, creating a sustainable national capacity for continuous quality improvement.
Central to these achievements was AFENET technical support delivered along two strategic lines of action: [1]. laboratory and clinical services—provided by a multidisciplinary team of experienced physicians and, [2]. bioanalysis, through continuous follow-up and evaluation visits. By adhering to international standards; targeted capacity building; and systematic documentation and sharing of best practices, we achieved significant progress as demonstrated by improved institutional collaboration and achievement of project goals.
A major lesson learnt from our experience implementing this initiative is the need for robust and resilient supply chains to ensure timely and traceable delivery of results, reinforced by comprehensive follow-up systems and long-term investment in human resources. These will remedy the observed challenges of intermittent supply of reagents for HIV viral load testing; shortage of specialized staff and suboptimum adherence by patients to prescribed regimens.
AFENET is committed to consolidating the gains by working with the Government and other partners to: extend ISO accreditation to all HIV viral load testing laboratories; secure sustainable financing; and achieve the UNAIDS 95-95-95 targets. We aspire to provide a robust and replicable model for HIV service delivery and laboratory excellence across the region and beyond.



